correct option is D
Explanation: There are 3 main types of bonding:
1) Ionic bond: It is a type of chemical bond that involves the complete transfer of valence electrons between atoms forming bond.
2) Metallic Bond: It is a type of chemical bond which is present between two metal elements.
3) Covalent Bond: It is a type of chemical bond which involves sharing of electron pairs between 2 atoms forming bond. This bonding is classified further into two types - Polar Covalent bond and Non-polar covalent bond.
Polar covalent bond: This bond is present between two atoms having different electronegativities and thus forming a dipole. For example, in HCl, the electronegativity of H is 2.1 and that of Cl is 3 thus, forming a polar covalent bond. Non-polar covalent bond: This bond is present between two atoms having same electronegativities and thus forming no dipole. For example, In
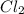
molecule Cl has a electronegativity of 3.0 and both the electronegativities gets cancelled out. Thus, forming a non-polar covalent bond.
In the question the bond is present between two chlorine atoms, therefore it is forming a non-polar covalent bond.


















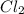 molecule Cl has a electronegativity of 3.0 and both the electronegativities gets cancelled out. Thus, forming a non-polar covalent bond.
molecule Cl has a electronegativity of 3.0 and both the electronegativities gets cancelled out. Thus, forming a non-polar covalent bond.




