
Chemistry, 24.07.2019 10:30, graymonky12
Balance the following equation. then determine the ratio for the products kcl and o2 generated during the decomposition of potassium chlorate. kclo3 kcl + o2 1: 1 2: 2 4: 3 2: 3 3: 2

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:00, bernicewhite156
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2


Do you know the correct answer?
Balance the following equation. then determine the ratio for the products kcl and o2 generated durin...
Questions in other subjects:




Computers and Technology, 18.02.2021 09:10

Mathematics, 18.02.2021 09:10


Biology, 18.02.2021 09:10



Physics, 18.02.2021 09:10


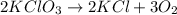
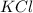 and 3 moles of
and 3 moles of 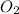 .
.



