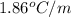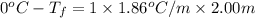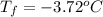Answers: 1
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 14:30, Kiaraboyd9366
Select all of the statements which are true. electrons are located in shells or orbits around the atom. electrons orbit slowly around the atom. electrons travel in one flat path around the nucleus of an atom. the valence of an atom is determined by the number of electrons in the atom's outermost shell.
Answers: 1
Do you know the correct answer?
Calculate the freezing point of a 2.00 molal solution of the nonelectrolyte glucose. the freezing po...
Questions in other subjects:











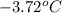
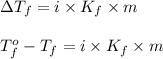
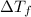 = change in freezing point
= change in freezing point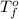 = temperature of pure water =
= temperature of pure water = 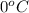
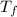 = temperature of solution = ?
= temperature of solution = ?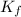 = freezing point constant =
= freezing point constant =