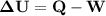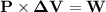An ideal gaseous reaction (which is a hypothetical gaseous reaction that conforms to the laws governing gas behavior) occurs at a constant pressure of 45.0 atm and releases 68.0 kj of heat. before the reaction, the volume of the system was 6.40 l . after the reaction, the volume of the system was 2.80 l . calculate the total internal energy change, δe, in kilojoules.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, hannah5143
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Do you know the correct answer?
An ideal gaseous reaction (which is a hypothetical gaseous reaction that conforms to the laws govern...
Questions in other subjects:

Mathematics, 10.06.2021 03:30

Biology, 10.06.2021 03:30


English, 10.06.2021 03:30

Mathematics, 10.06.2021 03:30

Social Studies, 10.06.2021 03:30

Mathematics, 10.06.2021 03:30

Mathematics, 10.06.2021 03:30