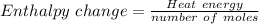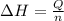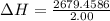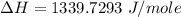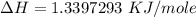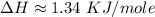Chemistry, 20.07.2019 13:20, thomasdianne
Atotal of 2.00 mol of a compound is allowed to react with water in a foam coffee cup and the reaction produces 173 g of solution. the reaction caused the temperature of the solution to rise from 21.00 to 24.70 ∘c. what is the enthalpy of this reaction? assume that no heat is lost to the surroundings or to the coffee cup itself and that the specific heat of the solution is the same as that of pure water. enter your answer in kilojoules per mole of compound to three significant figures.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, Bryanguzman2004
Water (2510 g ) is heated until it just begins to boil. if the water absorbs 5.09×105 j of heat in the process, what was the initial temperature of the water?
Answers: 3


Chemistry, 22.06.2019 20:30, trevorhenyan51
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1

Chemistry, 22.06.2019 22:10, steven0448
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1
Do you know the correct answer?
Atotal of 2.00 mol of a compound is allowed to react with water in a foam coffee cup and the reactio...
Questions in other subjects:






Mathematics, 12.05.2021 22:40

Mathematics, 12.05.2021 22:40


Mathematics, 12.05.2021 22:40