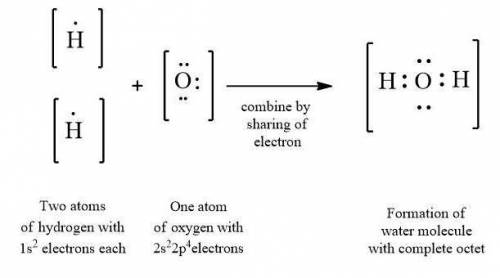
The octet rule defined as the rule according to that every atom has a tendency to acquire eight electrons in its outermost shell. The atom with incomplete octet leads to the formation of covalent or ionic compound to attain stability. For example, hydrogen and oxygen share electrons to complete their octet. (Refer to the attached image).
Further Explanation:
Octet rule states that for the stability of any element, it must have a valence shell of eight electrons. Octet means a group of eight and therefore for stability of any element it should have 8 electrons.
Atoms of same or different elements with an incomplete electronic configuration that is having electrons less than 8 are unstable and they combine together to form stable molecules with a complete octet.
For example, water molecule 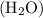 ,
,
In a water molecule, hydrogen(H) has an electronic configuration of 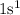 . Hydrogen requires one electron in its valence shell to complete its nearest noble gas configuration of helium(He) that is duplet (means group of 2).
. Hydrogen requires one electron in its valence shell to complete its nearest noble gas configuration of helium(He) that is duplet (means group of 2).
Whereas oxygen(O) has an electronic configuration of 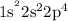 or
or ![\;\left[ {{\text{He}}} \right]2{{\text{s}}^2}2{{\text{p}}^4}](/tpl/images/0697/9933/c00fc.png) . Oxygen requires two electrons in its valence shell to reach its the nearest noble gas configuration of neon(Ne) that is octet.
. Oxygen requires two electrons in its valence shell to reach its the nearest noble gas configuration of neon(Ne) that is octet.
So for the stability, two atoms of hydrogen element share their one electron each with one atom of oxygen element, thereby resulting in a molecule of water with two covalent bonds. The formation of a water molecule leads to the completion of octet of both oxygen and hydrogen. (Refer to the attached image).
Learn more:
1. The octet rule:
2. How many covalent bond nitrogen forms:
Answer details:
Grade: Secondary School
Subject: Chemistry
Chapter: Chemical Bonding
Keywords: octet rule, noble gas, stability, hydrogen, oxygen, electronic configuration, covalent, sharing, water molecule and duplet.

















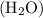 ,
,
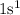 . Hydrogen requires one electron in its valence shell to complete its nearest noble gas configuration of helium(He) that is duplet (means group of 2).
. Hydrogen requires one electron in its valence shell to complete its nearest noble gas configuration of helium(He) that is duplet (means group of 2).
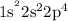 or
or ![\;\left[ {{\text{He}}} \right]2{{\text{s}}^2}2{{\text{p}}^4}](/tpl/images/0697/9933/c00fc.png) . Oxygen requires two electrons in its valence shell to reach its the nearest noble gas configuration of neon(Ne) that is octet.
. Oxygen requires two electrons in its valence shell to reach its the nearest noble gas configuration of neon(Ne) that is octet.





