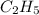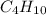Chemistry, 18.07.2019 00:00, hihowareyou12
Asample of a hydrocarbon contains 20.75 g c and 4.25 g h . it’s molar mass is 58.04 g/mol . what is its empirical formula

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 01:30, jusicca1109
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1


Chemistry, 22.06.2019 16:00, yfnal3x
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
Do you know the correct answer?
Asample of a hydrocarbon contains 20.75 g c and 4.25 g h . it’s molar mass is 58.04 g/mol . what is...
Questions in other subjects:

Biology, 25.08.2019 09:10

Mathematics, 25.08.2019 09:10






Mathematics, 25.08.2019 09:10

Biology, 25.08.2019 09:10