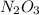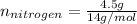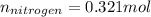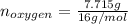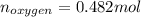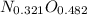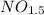Chemistry, 15.07.2019 06:30, sierravick123owr441
Acompound consists of 4.50 g of nitrogen for every 7.71 g of oxygen and has the same empirical and molecular formula identify the compound

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 08:00, wizz4865
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 13:10, bartonamber4042
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1
Do you know the correct answer?
Acompound consists of 4.50 g of nitrogen for every 7.71 g of oxygen and has the same empirical and m...
Questions in other subjects:



Mathematics, 24.11.2020 14:00


Mathematics, 24.11.2020 14:00


Mathematics, 24.11.2020 14:00

Mathematics, 24.11.2020 14:00