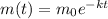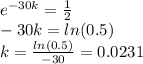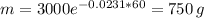Chemistry, 13.07.2019 19:30, kaytlynnmeyer
Anuclear waste site. cesium-137 is a particularly dangerous by-product of nuclear reactors. it has a half-life of 30 years. it can be readily absorbed into the food chain and is one of the materials that would be stored in the proposed waste site at yucca mountain (see the article opening this section). suppose we place 3000 grams of cesium-137 in a nuclear waste site. how much cesium-137 will be present after 60 years, or two half-lives?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:00, yaneiryx5476
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1

Chemistry, 22.06.2019 17:30, latezwardjr15
In a heat of an engine, if 700 j enters the system, and the piston does 400 j of work what is the final internal (thermal) energy of the system if the initial energy is 1,500 j
Answers: 2

Chemistry, 22.06.2019 20:10, jakhunter354
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3
Do you know the correct answer?
Anuclear waste site. cesium-137 is a particularly dangerous by-product of nuclear reactors. it has a...
Questions in other subjects:




Mathematics, 21.06.2021 20:20




Mathematics, 21.06.2021 20:20