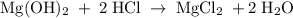Chemistry, 13.07.2019 08:40, yashirachevalier
A0.784 g sample of magnesium is added to a 250 ml flask and dissolved in 150ml of water. magnesium hydroxide obtained from the reaction required 215.0 ml of 0.300 m hydrochloric acid to completely react. how many moles of mg were used?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, hala201490
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 23.06.2019 06:10, tammydbrooks43
How much would the freezing point of water decrease if 4 mol of nacl were added to 1 kg of water (kf=1.86 degrees c/(mol/kg) for water and i=2 for nacl a- 7.44 degrees c b- 14.88 c 3.72 d 1.86
Answers: 1
Do you know the correct answer?
A0.784 g sample of magnesium is added to a 250 ml flask and dissolved in 150ml of water. magnesium h...
Questions in other subjects:



English, 13.07.2019 04:30

Mathematics, 13.07.2019 04:30

Social Studies, 13.07.2019 04:30

Social Studies, 13.07.2019 04:30

Social Studies, 13.07.2019 04:30



Social Studies, 13.07.2019 04:30


 .
.