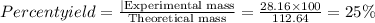Chemistry, 12.07.2019 21:40, jsharma57p7enrw
Give the percent yield when 28.16 g of co2 are formed from the reaction of 8.000 moles of c8h18 with 4.000 moles of o2. 2 c8h18 + 25 o2 → 16 co2 + 18 h2o

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:00, hannah2757
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1

Chemistry, 22.06.2019 22:00, aliciaa101
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1
Do you know the correct answer?
Give the percent yield when 28.16 g of co2 are formed from the reaction of 8.000 moles of c8h18 with...
Questions in other subjects:


Arts, 23.04.2021 20:40

Mathematics, 23.04.2021 20:40

Mathematics, 23.04.2021 20:40


Mathematics, 23.04.2021 20:40


Mathematics, 23.04.2021 20:40

Mathematics, 23.04.2021 20:40


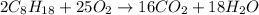
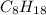 , then 4 moles of oxygen will react with
, then 4 moles of oxygen will react with 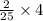 moles of
moles of 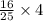 moles of carbon-dioxide gas that is 2.56 moles.
moles of carbon-dioxide gas that is 2.56 moles.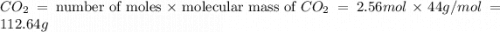
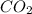 = 28.16 g
= 28.16 g