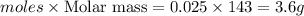The mass in grams of AgCl produced when 4.22 g of AgNO₃ react with 7.73 g of AlCl₃ is 3.56 g
From the question, the equation of the reaction is given as
3agno3 (aq) + alcl3 (aq) → al(no3)3 (aq) + 3agcl (s)
This can be written properly as
3AgNO₃ (aq) + AlCl₃ (aq) → Al(NO₃)₃ (aq) + 3AgCl (s)
From the balanced chemical equation above, we observe that
3 moles of AgNO₃ will react with 1 mole of AlCl₃ to yield 1 mole of Al(NO₃)₃ and 3 moles of AgCl.
To determine the mass of AgCl that would be produced when 4.22 g of AgNO₃ react with 7.73 g of AlCl₃,
First, we will convert the given masses to number of moles
Using the formula,
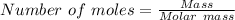
For AgNO₃
Mass = 4.22 g
Molar mass = 169.87 g/mol
∴ 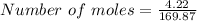
Number of moles of AgNO₃ present = 0.02484 moles
For AlCl₃
Mass = 7.73 g
Molar mass = 133.34 g/mol
∴ 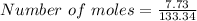
Number of moles of AlCl₃ present = 0.05797 moles
Since,
3 moles of AgNO₃ react with 1 mole of AlCl₃
Then,
0.02484 moles of AgNO₃ will react with 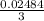 moles of AlCl₃ completely.
moles of AlCl₃ completely.
(NOTE: AgNO₃ is the limiting reagent and AlCl₃ is the excess reagent)
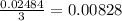
∴ Only 0.00828 moles of AlCl₃ will react.
Now, since
3 moles of AgNO₃ will react with 1 mole of AlCl₃ to yield 1 mole of Al(NO₃)₃ and 3 moles of 3AgCl
Therefore,
0.02484 moles of AgNO₃ will react with 0.00828 mole of AlCl₃ to yield 0.00828 mole of Al(NO₃)₃ and 0.02484 moles of AgCl
∴ 0.02484 moles of AgCl is produced during the reaction.
Now, we will convert this amount to grams
From the formula
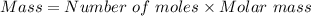
Number of moles = 0.02484 moles
Molar mass = 143.32 g/mol
∴ Mass of AgCl produced = 0.02484 × 143.32
Mass of AgCl produced = 3.56 g
Hence, the mass in grams of AgCl produced when 4.22 g of AgNO₃ react with 7.73 g of AlCl₃ is 3.56 g
Learn more here: link















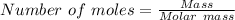
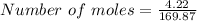
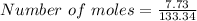
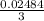 moles of AlCl₃ completely.
moles of AlCl₃ completely. 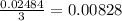
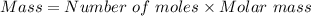
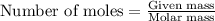
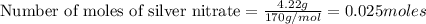
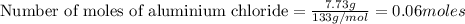
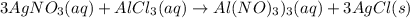
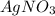 react with 1 mole of
react with 1 mole of 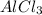
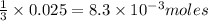 of
of 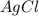
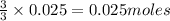 of
of