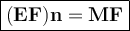The different ways to represent compounds:
a structural formula, an empirical formula, a space-filling model, a molecular formula and a ball stick model
Further explanation
The empirical formula (EF) is the smallest atomic ratio of the constituent elements.
The empirical formula shows the simplest comparison of the molecules making up the molecule
The Molecular formula (MF) is a formula that shows the number of atomic elements that make up a compound.
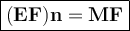
The empirical formula can be identical to the molecular formula. Ion compounds only have an empirical formula and do not have a molecular formula because ion compounds are formed from positive ions and negative ions, not from molecules.
Structural formulas are depictions of bonds between atoms in a molecule. The structural formula is obtained from the Lewis formula by replacing each pair of electrons with a line. From this structural formula, you can see a single bond or a double bond
A ball-and-stick model describes an atom as a ball and its bond as a stick
A space-filling molecular model describes molecules like a ball-and-stick model but without the stick
Whereas the Kolbe formula, a ball model, and a cylindric model are unknown
Learn more
chemical bond
link
ionic bonding
link
electron dots
link
Keywords: a structural formula, an empirical formula, a space-filling model, a molecular formula, a ball stick model