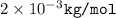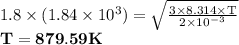Chemistry, 03.07.2019 22:00, codysaucier2823
The urms of h2 molecules at 273k is 1.84×103m/s. part aat what temperature is urms for h21.8 times greater than this value?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, elizabethprasad2
How many grams of n2h4 will be consumed by 23 g of n2o4
Answers: 1


Chemistry, 22.06.2019 03:50, daniel9299
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
Do you know the correct answer?
The urms of h2 molecules at 273k is 1.84×103m/s. part aat what temperature is urms for h21.8 times g...
Questions in other subjects:

Mathematics, 18.03.2021 20:40


Social Studies, 18.03.2021 20:40

Advanced Placement (AP), 18.03.2021 20:40

Spanish, 18.03.2021 20:40


Mathematics, 18.03.2021 20:40

English, 18.03.2021 20:40

Mathematics, 18.03.2021 20:40

Mathematics, 18.03.2021 20:40


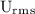 )
)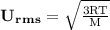
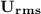 is Root Mean Square Velocity in m/s.
is Root Mean Square Velocity in m/s.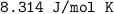
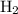 which is
which is