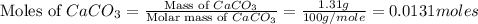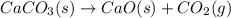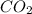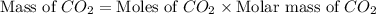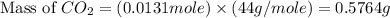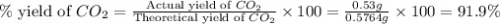Chemistry, 03.07.2019 22:00, laraekeyes
Asample of 0.53 g of carbon dioxide was obtained by heating 1.31 g of calcium carbonate. what is the percent yield for this reaction? caco3(s) ⟶ cao(s) + co2(s)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, carter1809
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 06:30, irvinbhangal2
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 10:30, ciel8809
Aglow stick contains a glass vial with chemicals. when the glow stick is bent, the vial breaks and the chemicals react to produce a glow. a science student observes that a glow stick kept in the freezer glows for a longer duration than a glow stick kept at room temperature. what conclusion can be drawn based on the observation? be sure to note the outcome and test variables in the conclusion.
Answers: 1
Do you know the correct answer?
Asample of 0.53 g of carbon dioxide was obtained by heating 1.31 g of calcium carbonate. what is the...
Questions in other subjects:

English, 17.06.2021 14:50

Mathematics, 17.06.2021 14:50

Social Studies, 17.06.2021 14:50

Business, 17.06.2021 14:50

Mathematics, 17.06.2021 14:50

Mathematics, 17.06.2021 14:50


Mathematics, 17.06.2021 14:50

History, 17.06.2021 14:50


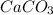 .
.