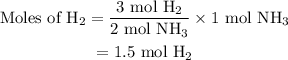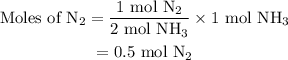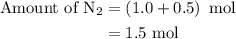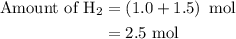Chemistry, 03.07.2019 16:30, campbell387
Amixture of n2(g) and h2(g) reacts in a closed container to form ammonia, nh3(g). the reaction ceases before either reactant has been totally consumed. at this stage 1.0 mol n2, 1.0 mol h2, and 1.0 mol nh3 are present. part a how many moles of n2 and h2 were present originally?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, shayneseaton
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1

Chemistry, 22.06.2019 12:30, Svetakotok
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Do you know the correct answer?
Amixture of n2(g) and h2(g) reacts in a closed container to form ammonia, nh3(g). the reaction cease...
Questions in other subjects:


English, 31.07.2019 12:20

Mathematics, 31.07.2019 12:20



Mathematics, 31.07.2019 12:20

Mathematics, 31.07.2019 12:20




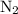 initially taken is
initially taken is 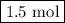 and the amount of
and the amount of 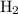 initially taken is
initially taken is 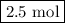 .
.
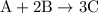
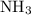 is as follows:
is as follows:
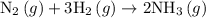
 .
.