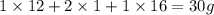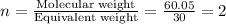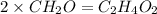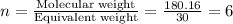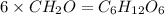The empirical formula for two compounds that have very different properties (one tastes very sour and the other very sweet) is ch2o. if the molar mass of compound a is 60.05 g/mol and compound b is 180.16 g/mol, what are the molecular formulas for these compounds, respectively?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, ashtonbillups
Is this a scientific model? use complete sentences to explain why or why not. a graphic organizer showing the water cycle
Answers: 3

Chemistry, 21.06.2019 18:00, peaceouthjkdrb2398
Rutherford's experiment indicated that matter was not as uniform as it appears what part of his experimental results implied this idea
Answers: 1

Chemistry, 22.06.2019 18:00, kingamir
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1
Do you know the correct answer?
The empirical formula for two compounds that have very different properties (one tastes very sour an...
Questions in other subjects:

Mathematics, 20.03.2020 12:05

Mathematics, 20.03.2020 12:05


Geography, 20.03.2020 12:05

Mathematics, 20.03.2020 12:05



Mathematics, 20.03.2020 12:05

Mathematics, 20.03.2020 12:05


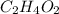
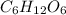
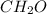 , empirical weight is =
, empirical weight is =