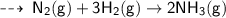Chemistry, 30.01.2022 01:00, SKYBLUE1015
The balanced chemical equation for the reaction of nitrogen with hydrogen to produce
Ammonia gas is-
A. N2(g) + H2(g) → 2NH3(g) B. 2N2(g) + H2(g) → 2NH3(g)
C. N2(g) + 3H2(g) → 2NH3(g) D. 2N2(g) + H2(g) →2NH3(g)

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 20:30, mayamabjishovrvq9
1. calculate the approximate enthalpy of the reaction in joules. estimate that 1.0 ml of vinegar has the same thermal mass as 1.0 ml of water. iqnore the thermal mass of th sodium bicarbonate. note: it takes about 4.2 joules () to change 1.0 gram (1.0ml) of water 1.0 c
Answers: 2

Chemistry, 22.06.2019 04:00, nothingworksoutforme
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 09:00, mercymain1014
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1
Do you know the correct answer?
The balanced chemical equation for the reaction of nitrogen with hydrogen to produce
Ammonia gas i...
Questions in other subjects:

English, 09.03.2021 19:40



Mathematics, 09.03.2021 19:40


Mathematics, 09.03.2021 19:40

Chemistry, 09.03.2021 19:40

Mathematics, 09.03.2021 19:40

Mathematics, 09.03.2021 19:40

History, 09.03.2021 19:40