Chemistry, 27.01.2022 14:00, Svetakotok
Sulfur trioxide decomposes to sulfur dioxide and oxygen
gas as shown in the following reaction:
2503 (g) 2502 (g) + O2 (g)
Given a 240.0 g sample of sulfur trioxide (MM = 80.1 g/mol),
how many grams of oxygen (MM = 32.0 g/mol) are
produced, assuming the decomposition goes to
completion?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, hannah5143
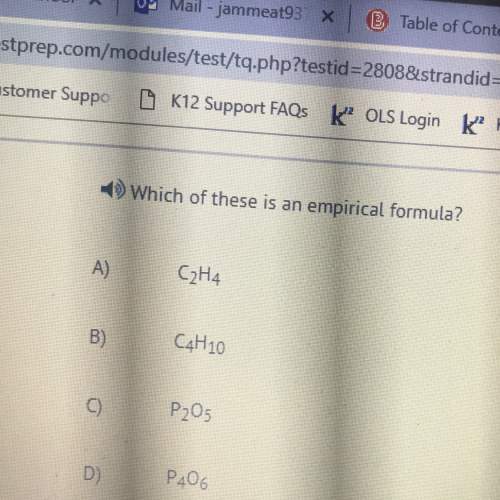
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 21.06.2019 21:40, MathChic68
It is important to wear proper protective equipment in lab even when not actively performing experiments because accidents can affect any researcher, even one not working on an experiment. select the best answer from the choices provided
Answers: 3

Chemistry, 21.06.2019 23:30, ashleyjaslin
Calculate the expected ph values of the buffer systems from the experiments (a, b,c, d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2
Do you know the correct answer?
Sulfur trioxide decomposes to sulfur dioxide and oxygen
gas as shown in the following reaction:
Questions in other subjects:



Mathematics, 26.04.2021 23:30

Mathematics, 26.04.2021 23:30



Biology, 26.04.2021 23:30


History, 26.04.2021 23:30