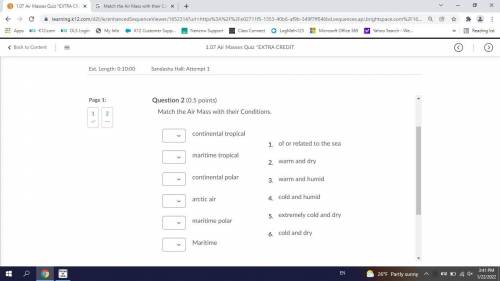
Can someone answer these please i'll give 25 points it would mean alot to me <3
...


Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, acaciacoats
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2



Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Geography, 18.11.2020 23:00

History, 18.11.2020 23:00

English, 18.11.2020 23:00

Engineering, 18.11.2020 23:00

History, 18.11.2020 23:00



Mathematics, 18.11.2020 23:00

English, 18.11.2020 23:00