
Chemistry, 22.01.2022 06:50, michaelwarren8728
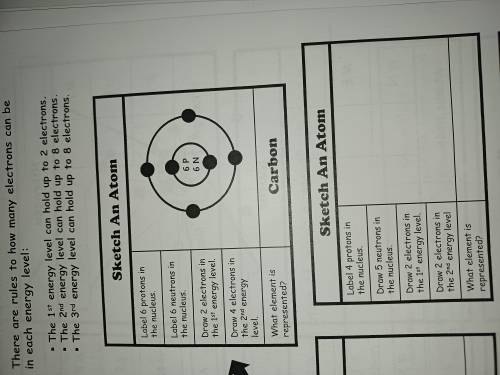
Pleasee do the attachment quickly it's due btw the top one is the example just tell me how to sketch the bottom one

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 22:40, lindseyklewis1p56uvi
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization. a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution. part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 05:00, mprjug6
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 4.20 mol fe and 6.70 mol nio(oh) react?
Answers: 3
Do you know the correct answer?
Pleasee do the attachment quickly it's due btw the top one is the example just tell me how to sketch...
Questions in other subjects:


History, 19.01.2020 11:31


Mathematics, 19.01.2020 11:31


Biology, 19.01.2020 11:31

History, 19.01.2020 11:31

Mathematics, 19.01.2020 11:31







