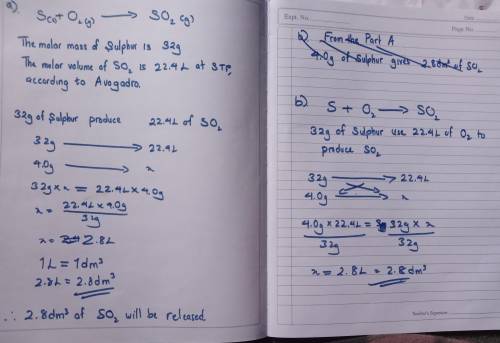Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, emfranco1
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2

Chemistry, 22.06.2019 15:30, 20cschultz
Which suspect most likely committed the robbery and how do you know
Answers: 2

Chemistry, 22.06.2019 17:30, shookiegriffin
I'm learning about the periodic tables and what each subject's configuration is. for example, hydrogen is 1s^1, but i don't understand how you get that. can someone me understand how to figure out how to figure this out? sorry if the question makes no sense, but it would really a lot if you could me understand! you so much if you can!
Answers: 1
Do you know the correct answer?
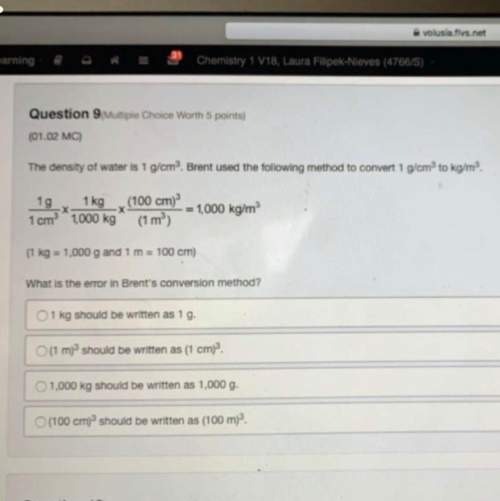
exactly 4.0g of sulfur is burnt in a fume extraction cupboard. The sulfur is ignited in an excess of...
Questions in other subjects:


Mathematics, 09.04.2020 07:31


Chemistry, 09.04.2020 07:31

Mathematics, 09.04.2020 07:31

Mathematics, 09.04.2020 07:31


English, 09.04.2020 07:31

Mathematics, 09.04.2020 07:31