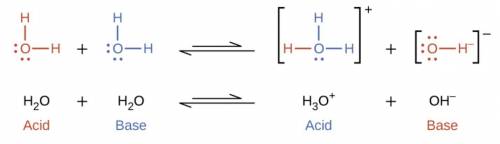
Which statement best describes the pH of pure water? It is neutral because the concentration of hydronium ions equals that of hydroxide ions. It is neutral because the pure liquid contains neither hydronium ions nor hydroxide ions. It is acidic because it has a hydronium ion concentration of mc027-1. Jpg. It is basic because it has a hydroxide ion concentration of mc027-2. Jpg.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 19:20, choiboiqg5755
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1

Chemistry, 22.06.2019 23:00, brianfranklin17
What is the correct lewis dot structure for arsenic?
Answers: 2
Do you know the correct answer?
Which statement best describes the pH of pure water? It is neutral because the concentration of hydr...
Questions in other subjects: