
Chemistry, 26.12.2021 04:10, bobbyhill24
If one gram of sulphur dioxide contains x molecules what will be the number of molecules in 1g of methane

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, jusicca1109
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1


Chemistry, 22.06.2019 10:10, jojomgarcia01
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 12:00, Alexislol7908
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1
Do you know the correct answer?
If one gram of sulphur dioxide contains x molecules what will be the number of molecules in 1g of me...
Questions in other subjects:

History, 08.10.2019 11:50

Geography, 08.10.2019 11:50



History, 08.10.2019 11:50

History, 08.10.2019 11:50

History, 08.10.2019 11:50

Mathematics, 08.10.2019 11:50

Mathematics, 08.10.2019 11:50



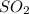
 mole of
mole of 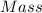
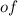
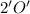
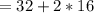
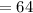 grams
grams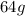 of sulphur dioxide
of sulphur dioxide 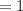 mole
mole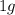 of sulphur dioxide =
of sulphur dioxide = 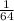 mole
mole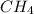
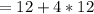
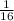 mole
mole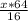 molecules
molecules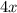 molecules
molecules




