
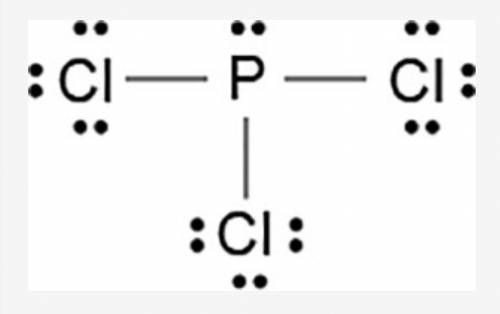
The Lewis dot model of a molecule is shown.
A visual diagram of a PCl3 molecule is shown. Phosphorous is the central atom with a horizontal line connecting to each of the three Chlorine atoms around it. Phosphorous has a pair of dots on it. Each of the three chlorine atoms have a pair of three dots on it.
Based on the model, which of the following is true?
The electronegativity difference between phosphorous and chlorine is greater than 1.7.
Each chlorine has three non-bonded pairs and one bonded pair of electrons.
Phosphorous has three non-bonded pairs and one bonded pair of electrons.
Phosphorous has three valence electrons in the outermost energy level.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, alfarodougoy8lvt
Agas is contained in a thick walled balloon when the pressure changes from 1.21 atm to 2.52 the volume changes from 3.75 l to 1.72 l and the temperature change from 293k to blank k
Answers: 3


Chemistry, 22.06.2019 11:00, hannah5143
The human eye contains a molecule called 11-cis-retinal that changes shape when struck with light of sufficient energy. the change in shape triggers a series of events that results in an electrical signal being sent to the brain that results in vision. the minimum energy required to change the conformation of 11-cis-retinal within the eye is about 164 kj/mol.
Answers: 2

Do you know the correct answer?
The Lewis dot model of a molecule is shown.
A visual diagram of a PCl3 molecule is shown. Phosphor...
Questions in other subjects:

Mathematics, 10.06.2020 04:57

Mathematics, 10.06.2020 04:57


Mathematics, 10.06.2020 04:57

Mathematics, 10.06.2020 04:57


Spanish, 10.06.2020 04:57








