
Chemistry, 17.12.2021 01:40, urfavgringo
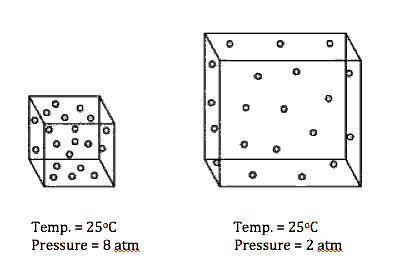
Based on the model, which conclusion can be made?
A) As volume increases, pressure decreases.
B) As volume increases, pressure increases.
C) Volume increases as temperature increases.
D) Density decreases as temperature decreases.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, sammysosa121832
The ph of carrots are 5.0 how it is classified a. acidic b. basic c. indicator d. neutral
Answers: 2


Chemistry, 22.06.2019 06:30, backup5485
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1
Do you know the correct answer?
Based on the model, which conclusion can be made?
A) As volume increases, pressure decreases.
Questions in other subjects:

English, 23.06.2019 11:00

Chemistry, 23.06.2019 11:00

Biology, 23.06.2019 11:00


Mathematics, 23.06.2019 11:00

Mathematics, 23.06.2019 11:00

Biology, 23.06.2019 11:00


Social Studies, 23.06.2019 11:00






