Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 17:20, banna01man
Pegmatites are igneous rocks in which the individual minerals are very large. typically, the minerals are all light-colored quartz, feldspar and muscovite. if you were given a black and white photograph of a pegmatite in a quarry (where the rock has been blasted and broken), what physical properties could you use to identify those three minerals in this hypothetical photo? describe each mineral and the specific diagnostic properties. be specific.
Answers: 2
Do you know the correct answer?
-
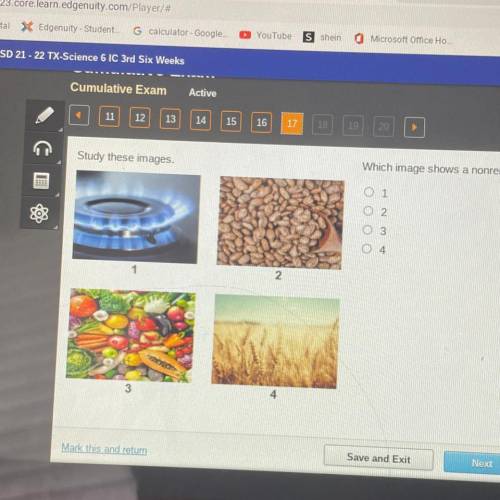
Which image shows in nonrenewable resource?
...
Which image shows in nonrenewable resource?
...
Questions in other subjects:


History, 16.10.2020 05:01

Biology, 16.10.2020 05:01


Mathematics, 16.10.2020 05:01

History, 16.10.2020 05:01

Arts, 16.10.2020 05:01

Business, 16.10.2020 05:01