
Chemistry, 09.12.2021 19:40, 2337911726
Consider the equations below.
4 equations. 1, Upper C Upper H Subscript 4 Baseline (g) right arrow Upper C (s) + 2 Upper H Subscript 2 Baseline (g) Delta H Subscript 1 Baseline = 74.6 kilojoules. 2, Upper C (s) + 2 Upper C l Subscript 2 Baseline (g) right arrow Upper C Upper Cl Subscript 4 Baseline (g) Delta H Subscript 2 Baseline = negative 95.7 kilojoules. 3, 2 Upper H Subscript 2 Baseline (g) + 2 Upper C l Subscript 2 Baseline (g) right arrow 4 Upper H Upper Cl (g) delta H Subscript 3 Baseline = negative 284.6 kilojoules. 4, Upper C Upper H Subscript 4 Baseline (g) + 4 Upper C l Subscript 2 Baselines (g) right arrow Upper C Upper C l Subscript 4 Baseline (g) + 4 Upper H Upper C L (g) Delta H 4 = negative 205.7 kilojoules.
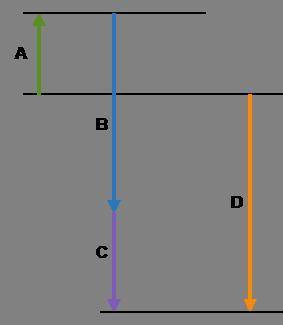
Complete the following based on the diagram.
Arrow A:
Arrow B:
Arrow C:
Arrow D:

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, tjjjjjjjjjjjjjjjjjjj
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1


Do you know the correct answer?
Consider the equations below.
4 equations. 1, Upper C Upper H Subscript 4 Baseline (g) right arrow...
Questions in other subjects:

Mathematics, 24.03.2021 21:30

History, 24.03.2021 21:30


Mathematics, 24.03.2021 21:30




Mathematics, 24.03.2021 21:30








