
Chemistry, 09.12.2021 19:30, djmelodiedaniels
Consider the equations below.
4 equations. 1, Upper C Upper H Subscript 4 Baseline (g) right arrow Upper C (s) + 2 Upper H Subscript 2 Baseline (g) Delta H Subscript 1 Baseline = 74.6 kilojoules. 2, Upper C (s) + 2 Upper C l Subscript 2 Baseline (g) right arrow Upper C Upper Cl Subscript 4 Baseline (g) Delta H Subscript 2 Baseline = negative 95.7 kilojoules. 3, 2 Upper H Subscript 2 Baseline (g) + 2 Upper C l Subscript 2 Baseline (g) right arrow 4 Upper H Upper Cl (g) delta H Subscript 3 Baseline = negative 284.6 kilojoules. 4, Upper C Upper H Subscript 4 Baseline (g) + 4 Upper C l Subscript 2 Baselines (g) right arrow Upper C Upper C l Subscript 4 Baseline (g) + 4 Upper H Upper C L (g) Delta H 4 = negative 205.7 kilojoules.
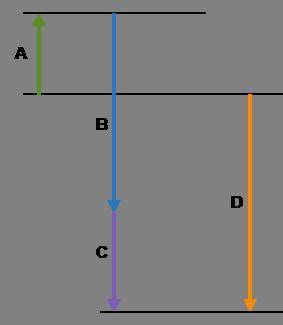
Complete the following based on the diagram.
Arrow A:
Arrow B:
Arrow C:
Arrow D:

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 15:30, kfull6027
The amount of iron in ore can be quantitatively determined by titrating a solution of the unknown with a standard solution of dichromate, cr2o72−. the net ionic equation is 6fe2+(aq)+cr2o72−(aq)+14h+(aq)→6fe3 +(aq)+2cr3+(aq)+7h2o(aq) part a the titration of 25.0 ml of an iron(ii) solution required 18.0 ml of a 0.230 m solution of dichromate to reach the equivalence point. what is the molarity of the iron(ii) solution?
Answers: 1


Chemistry, 23.06.2019 19:00, tiffanibell71
Which law was used to determine the relationship between the volume and the number of moles in this equation
Answers: 2

Chemistry, 23.06.2019 21:20, robert7248
A. explain, in terms of particles, why nacl(s) does not conduct electricity.
Answers: 1
Do you know the correct answer?
Consider the equations below.
4 equations. 1, Upper C Upper H Subscript 4 Baseline (g) right arrow...
Questions in other subjects:

Mathematics, 27.08.2021 07:10


History, 27.08.2021 07:10


Mathematics, 27.08.2021 07:10



English, 27.08.2021 07:10

History, 27.08.2021 07:10

Physics, 27.08.2021 07:20






