Chemistry, 08.12.2021 20:40, lizzie3545
the pressure exerted on a sample of a fixed amount of gas is doubled at constant temperature, and then the temperature of the gas in kelvins is doubled at constant pressure. what is the final volume of the gas?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:00, tahjaybenloss16
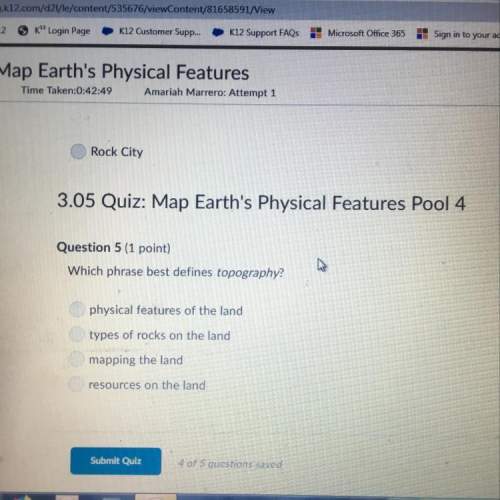
The pressure inside a hydrogen-filled container was 2.10 atm at 21 ? c. what would the pressure be if the container was heated to 92 ? c ?
Answers: 2

Chemistry, 22.06.2019 02:50, giiffnlojd
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3
Do you know the correct answer?
the pressure exerted on a sample of a fixed amount of gas is doubled at constant temperature, and th...
Questions in other subjects:

Physics, 27.11.2019 07:31

Mathematics, 27.11.2019 07:31




Mathematics, 27.11.2019 07:31

Mathematics, 27.11.2019 07:31

History, 27.11.2019 07:31


Mathematics, 27.11.2019 07:31