Mass of zinc reacts when 8.45 moles of HCl react to produce zinc chloride?
Zn+ 2HCl ZnCl+H2...

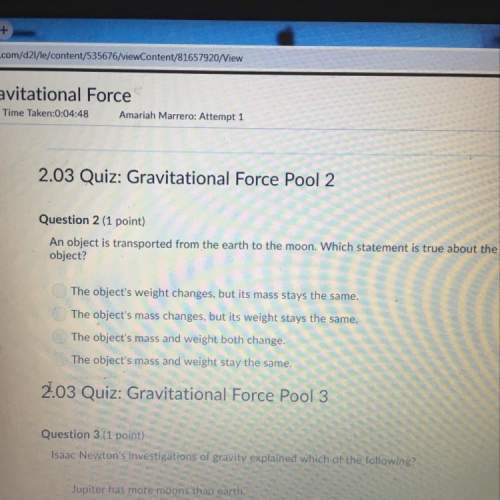
Chemistry, 02.12.2021 19:40, karlrey4401
Mass of zinc reacts when 8.45 moles of HCl react to produce zinc chloride?
Zn+ 2HCl ZnCl+H2

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:50, rebeccamckellpidge
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 19:50, ellycleland16
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 22.06.2019 20:20, catchonyet
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1

Chemistry, 22.06.2019 21:20, skyemichellec
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3
Do you know the correct answer?
Questions in other subjects:

History, 28.07.2021 07:10

Mathematics, 28.07.2021 07:10

Mathematics, 28.07.2021 07:10

English, 28.07.2021 07:10

Social Studies, 28.07.2021 07:10