
Chemistry, 22.11.2021 23:50, sadeed00974
10.00g of hydrated sodium sulfate decomposes to form 4.40g Na(2)SO(4).xH(2)O -> NaSO(4) + xH(2)O of anhydrous sodium sulfate on heating. What’s the formula mass of hydrated sodium sulfate and the value of x? please help i have no clue!
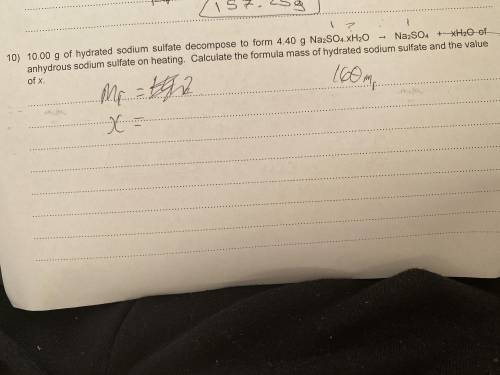

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 10:40, 1r32tgy5hk7
Question 17 hydrogen is manufactured on an industrial scale by this sequence of reactions: +ch4gh2og ⇌ +cog3h2g k1 +cogh2og ⇌ +co2gh2g k2 the net reaction is: +ch4g2h2og ⇌ +co2g4h2g k write an equation that gives the overall equilibrium constant k in terms of the equilibrium constants k1 and k2. if you need to include any physical constants, be sure you use their standard symbols, which you'll find in the aleks calculator.
Answers: 2
Do you know the correct answer?
10.00g of hydrated sodium sulfate decomposes to form 4.40g Na(2)SO(4).xH(2)O -> NaSO(4) + xH(2)O...
Questions in other subjects:



Mathematics, 14.08.2020 06:01

Mathematics, 14.08.2020 06:01

Mathematics, 14.08.2020 06:01

Geography, 14.08.2020 06:01


History, 14.08.2020 06:01


Mathematics, 14.08.2020 06:01







