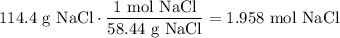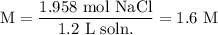Chemistry, 24.10.2021 19:10, coollid876
What is the molarity of a solution that contains 114.4g NaCl (molar mass= 58.44 g/mol) in 1.2 L solution?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, nothingworksoutforme
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 08:40, kellymcdow5135
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 17:40, Snowball080717
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3
Do you know the correct answer?
What is the molarity of a solution that contains 114.4g NaCl (molar mass= 58.44 g/mol) in 1.2 L solu...
Questions in other subjects:

Mathematics, 29.08.2019 19:30


Health, 29.08.2019 19:30


History, 29.08.2019 19:30




Mathematics, 29.08.2019 19:30