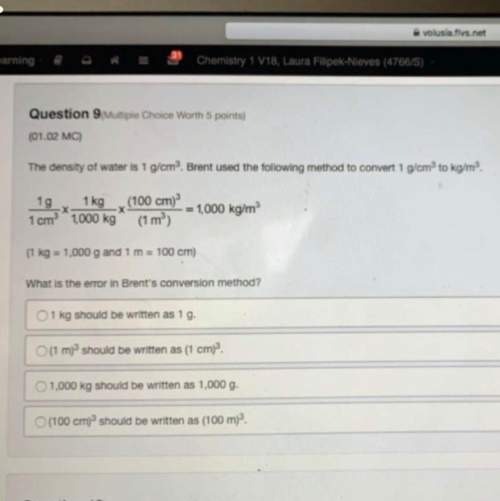
Chemistry, 11.10.2021 14:00, isaicruz2018
Calculate ΔH for the following reaction:
PCl3 (l) + Cl2 (g) → PCl5 (s)
given the following:
1) 2 P (s) + 3 Cl2 (g) → 2 PCl3 (l) ΔH = -639.4 kJ
2) 2 P (s) + 5 Cl2 (g) → 2 PCl5 (s) ΔH = -887.0 kJ

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 01:30, emfranco1
Which of the following statements is true about energy quantization at the atomic level? electrons in the outermost orbits are the most stable. electrons in all the orbits around the nucleus have the same amount of energy. electrons in the orbit closest to the nucleus have the least amount of energy. electrons absorb or release the same amount of energy independent of the energy levels.
Answers: 1


Chemistry, 23.06.2019 09:30, oscarruiz
The allotropes of carbon include a variety of structures that include three-dimensional tetrahedral lattices, planes of hexagonal rings, cylindrical tubes of hexagonal rings, and spheres of five- and six-membered rings. similar shapes of network covalent atomic solids are possible with carbon nitride, boron, and pure silicon (e. g., silicene is a graphene-like allotrope of pure silicon). in contrast, silicates exist as either highly ordered or amorphous (more random) three-dimensional lattices. what could explain why there are there no naturally occurring sheets, stacked sheets, cylindrical tubes, or spheres of network covalent atomic solids composed of silicon and oxygen (sio2)? would pure silicate structures make good lubricants or good electrical conductors?
Answers: 3

Chemistry, 23.06.2019 09:30, Dariana123
Which of the following is not a characteristic of a hydrogen bond? 1. it is responsible for the unusual physical properties of water. 2. it is weaker than a covalent bond. 3. it is stronger than other dipole-dipole interactions. 4. it can occur when hydrogen is covalently bound to very electronegative elements liks f, cl, br and i.
Answers: 1
Do you know the correct answer?
Calculate ΔH for the following reaction:
PCl3 (l) + Cl2 (g) → PCl5 (s)
given the follow...
given the follow...
Questions in other subjects:

Biology, 17.07.2019 13:50

Computers and Technology, 17.07.2019 13:50


Computers and Technology, 17.07.2019 13:50



Chemistry, 17.07.2019 13:50

Social Studies, 17.07.2019 13:50

Business, 17.07.2019 13:50

Biology, 17.07.2019 13:50