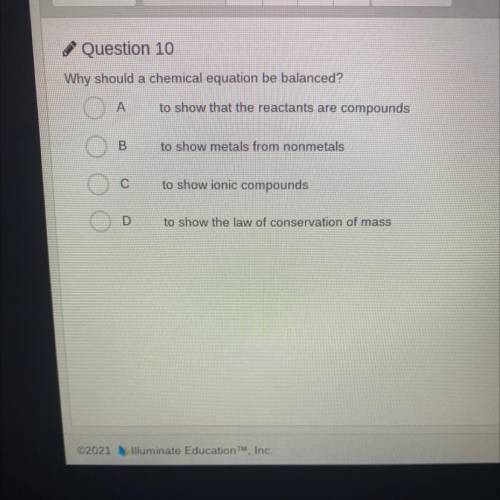
Why should a chemical equation be balanced?
A. To show that the reactants are compounds
B. T...

Chemistry, 07.10.2021 21:10, Zachary429
Why should a chemical equation be balanced?
A. To show that the reactants are compounds
B. To show metals from nonmetals
C. To show ionic compounds
D. To show the law of conservation of mass
Please answer no links please

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:50, karlyisaunicorn
Which real-world scenarios below represent physical and chemical changes? -running a car -exploding fireworks -mixing water and powdered drink mix -combining oil and vinegar to make salad dressing -taking aspirin for a headache -diluting bleach with water-digesting dinner-spreading peanut butter on bread
Answers: 2


Chemistry, 22.06.2019 10:00, valdezlizbeth6652
Why is carbon ideal for making different compounds?
Answers: 2

Chemistry, 22.06.2019 13:00, nadikadiaz1
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Physics, 22.12.2021 06:30




Social Studies, 22.12.2021 06:40



English, 22.12.2021 06:40


SAT, 22.12.2021 06:40






