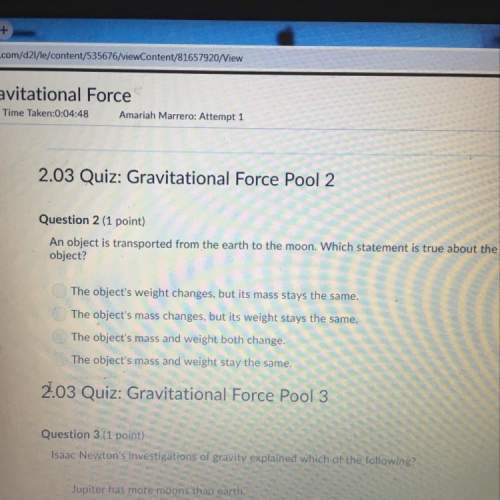
Chemistry, 07.10.2021 01:00, clevens123
The gas phase reaction of carbon monoxide with nitrogen dioxide is believed to occur by the following mechanism: step 1: 2 NO2 NO3 NO step 2: NO3 CO NO2 CO2 (a) Identify the molecularity of each step in the mechanism. step 1 unimolecular step 2 bimolecular (b) Write the equation for the net reaction. Use the smallest integer coefficients possible. If a box is not needed, leave it blank. SubScriptSuperScript Help SubScriptSuperScript Help SubScriptSuperScript Help SubScriptSuperScript Help (c) Identify any intemediates and/or catalysts in this mechanism. Catalyst: Enter formula. If none, leave box blank: SubScriptSuperScript Help Intermediate: Enter formula. If none, leave box blank: SubScriptSuperScript Help

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 18:00, Queenquestion5967
When the following equation is balanced using the smallest possible integers, what is the coefficent of oxygen gas? c7h16o(g) + o2(g) → co2(g) + h2o(g) -1 -5 -8 -16 -21
Answers: 3

Chemistry, 22.06.2019 21:30, leenzazou587
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
Do you know the correct answer?
The gas phase reaction of carbon monoxide with nitrogen dioxide is believed to occur by the followin...
Questions in other subjects:



English, 20.05.2021 02:10



History, 20.05.2021 02:10

English, 20.05.2021 02:10


Mathematics, 20.05.2021 02:10