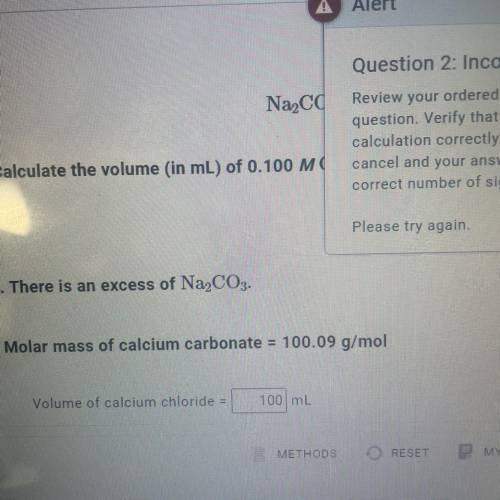
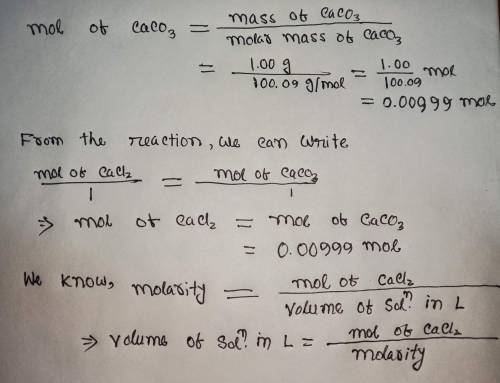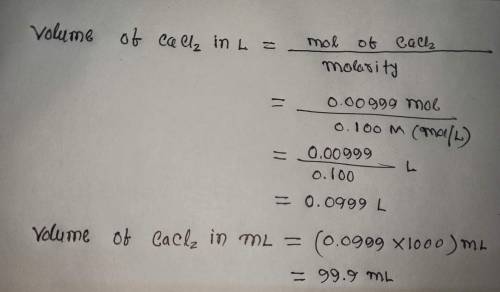Calculate the volume (in mL) of 0.100 M Na, CO3 needed to produce 1.00 g of
CaCO3(s)
. There...

Chemistry, 28.09.2021 03:10, hammackkatelyn60
Calculate the volume (in mL) of 0.100 M Na, CO3 needed to produce 1.00 g of
CaCO3(s)
. There is an excess of CaCl2.
Molar mass of calcium carbonate = 100.09 g/mol
*The answer is not 100ml or 10ml. Somehow the rounding up is not working well.*

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 16:00, krojas015
1. an experiment in your science class lists the materials needed for the lab. it is your job, as a lab partner, to measure out 25 ml of distilled water and 2.5 grams of magnesium. what lab measuring tools would you choose to measure each substance and how would you use each tool to get the correct amounts? be sure to describe the process you would follow step-by-step. (5 points) 2.which of the following is an si base unit for measuring mass? (2 points) ampere gram meter pound 3.which of the following is an si base unit for time? (2 points) decades hours minutes seconds 4.which of the following tools should a scientist use to measure an object in milligrams? (2 points) graduated cylinder pan balance tape measure thermometer 4.which of the following tools should a scientist use to measure an object in milligrams? (2 points) graduated cylinder pan balance tape measure thermometer. 5.a pencil beside a metric ruler. the ruler is scaled from 1 centimeter to 10 centimeters, with markings for millimeters between each number. one end of the pencil is beside the 0 on the ruler, and the pencil point is beside the 5. which of the following measurements is accurate but not precise? (2 points) 5 mm 5 cm 50 mm 50 cm 6. which of the following prefixes represents the largest value? (2 points) giga hector kilo milli 7. which of the following types of graphs is best for plotting the percentages of a whole value in a data set? (2 points) bar graph circle graph histogram line graph
Answers: 1


Chemistry, 23.06.2019 00:00, queenpaige2015
Which samples do the atoms have the least kinetic energy
Answers: 2

Chemistry, 23.06.2019 01:50, UncleVictor5188
Ablock of aluminum is dropped into a graduated cylinder with an initial volume of water at 75ml and the volumes rises to 90ml. if the block has a mass of 40.5 g what is its density ?
Answers: 1
Do you know the correct answer?
Questions in other subjects:





Spanish, 23.05.2020 23:02

Business, 23.05.2020 23:02

English, 23.05.2020 23:02

English, 23.05.2020 23:02

History, 23.05.2020 23:02

Mathematics, 23.05.2020 23:02