
Chemistry, 26.09.2021 01:40, VelvetXace
PLZ HELP :,I The density of an object is its mass per unit of volume. Calculate density by dividing each objects mass by its volume, and record the answers in the last column of your data table labeled Density. Remember the formula for Density! (hint: No naked numbers – label!)
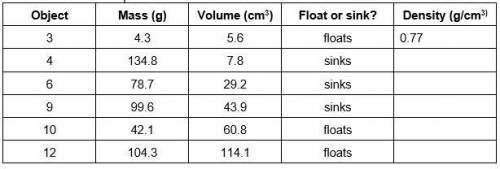

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, shafferakr6
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3


Chemistry, 22.06.2019 15:10, strodersage
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1

Chemistry, 22.06.2019 22:00, notearslefttocry14
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2
Do you know the correct answer?
PLZ HELP :,I
The density of an object is its mass per unit of volume. Calculate density by dividin...
Questions in other subjects:

History, 21.10.2020 14:01

Mathematics, 21.10.2020 14:01

Chemistry, 21.10.2020 14:01


Chemistry, 21.10.2020 14:01

Social Studies, 21.10.2020 14:01

English, 21.10.2020 14:01



Biology, 21.10.2020 14:01






