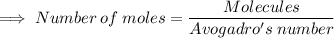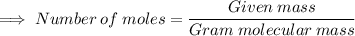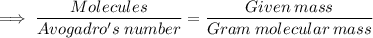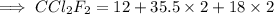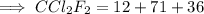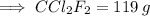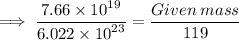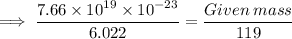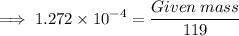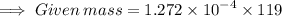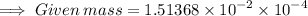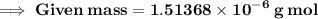Chemistry, 22.09.2021 14:00, Thelazysandwich
Calculate the mass (in grams) of 7.66×1019 CCl2F2 molecules

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, annsmith66
What is the result of multiplying (2.5 × 1010) × (2.0 × 10-7)? a. 5.0 × 103 b. 5.0 × 10-3 c. 5.0 × 1017 d. 5.0 × 10-17
Answers: 1

Chemistry, 22.06.2019 22:40, lindseyklewis1p56uvi
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization. a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution. part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 22.06.2019 23:00, catdog5225
What is formed when amino acids form long chains or polymerize
Answers: 1

Chemistry, 23.06.2019 03:30, uniqueray33
The molar mass of nickel(ni) is 58.7 g/mol. how many moles are in an 88 gram sample of nickel?
Answers: 1
Do you know the correct answer?
Calculate the mass (in grams) of 7.66×1019 CCl2F2 molecules...
Questions in other subjects:

Geography, 01.12.2020 19:30


Mathematics, 01.12.2020 19:30


History, 01.12.2020 19:30

Mathematics, 01.12.2020 19:30

English, 01.12.2020 19:30


Chemistry, 01.12.2020 19:30

History, 01.12.2020 19:30