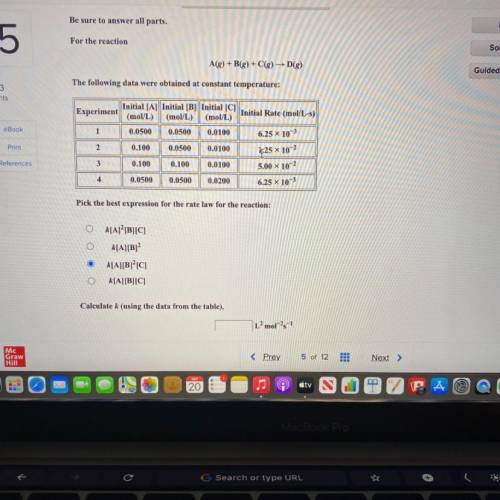
Be sure to answer all parts.
For the reaction
A(g) +B(g) + C(g) D()
The following data...

Be sure to answer all parts.
For the reaction
A(g) +B(g) + C(g) D()
The following data were obtained at constant temperature:
Initial [A] Initial [B] Initial [C]
Experiment
Initial Rate (mol/L-s)
(mol/L) (mol/L) (mol/L)
1 0.0500 0.0500 0.0100
6.25 x 103
2 0.100 0.0500 0.0100
3 0.100 0.100 0.0100 5.00 x 10-2
4 0.0500 0.0500 0.0200
6.25 x 10-3
1,25 x 10-2
Pick the best expression for the rate law for the reaction:
k[A] [B][C]
k[A][B]
k[A][B]°C]
k[A][B][C]
Calculate k (using the data from the table),
L’mol-1

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, melidacampos12
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2


Chemistry, 23.06.2019 03:00, rhianna18
In november 1987, a massive iceberg broke loose from the antartic ice mass and floated free in the ocean. the chunk of ice was estimated to be 98 mi long, 25 mi wide, and 750 ft thick. a typical backyard swimming pool contains about 24,000 gallons of water. how many of these pools could you fill from the water in this iceberg? (assume the iceberg is a rectangular solid of the above dimensions and consists of water only). express answer in scientific notation.
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Mathematics, 31.03.2020 20:54

History, 31.03.2020 20:54


Mathematics, 31.03.2020 20:54

History, 31.03.2020 20:54











