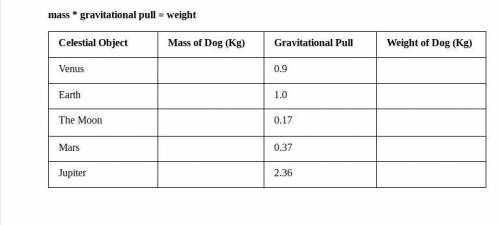
Hey i need help, questions are in image. It's due at 12pm mountain daylight time
...


Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, alexandroperez13
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 22.06.2019 06:00, momof7hardings
When would a bouncy ball have the most potential energy
Answers: 2


Chemistry, 22.06.2019 17:40, aaliyahthomas37
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1
Do you know the correct answer?
Questions in other subjects:


Mathematics, 14.12.2021 01:40

Mathematics, 14.12.2021 01:40

History, 14.12.2021 01:40

History, 14.12.2021 01:40





Mathematics, 14.12.2021 01:40