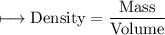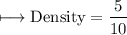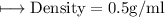Chemistry, 18.09.2021 06:40, rivasalejandro854
A vessel contains 30 mL of water. A sample of 5.0 g of copper metal is dropped into this vessel, raising the level of water in it to 40 mL. What is the density of the copper sample? Please show all steps leading to the final answer.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:00, SpiritedAway7087
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3

Chemistry, 23.06.2019 04:40, laurabwhiddon
Equal numbers of moles of he(g), ar(g), and ne(g) are placed in a glass vessel at room temperature. if the vessel has a pinhole-sized leak, which of the following will be true regarding the relative values of the partial pressures of the gases remaining in the vessel after some of the gas mixture has effused?
Answers: 1

Chemistry, 23.06.2019 06:00, irenecupcake4348
What does it mean for something to be dissolved in watera- it is submerged in water moleculesb-it is stirred in the water moleculesc- it is surrounded by water molecules d-it has water molecules added to it
Answers: 2
Do you know the correct answer?
A vessel contains 30 mL of water. A sample of 5.0 g of copper metal is dropped into this vessel, rai...
Questions in other subjects:




Physics, 29.04.2021 16:50






Mathematics, 29.04.2021 16:50