
Chemistry, 23.08.2021 23:30, ThousandSeas9381
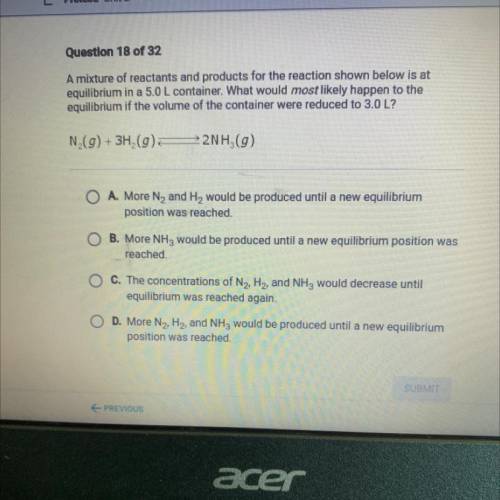
A mixture of reactants and products for the reaction shown below is at
equilibrium in a 5.0 L container. What would most likely happen to the
equilibrium if the volume of the container were reduced to 3.0 L?
N (g) + 3H2(g)
22NH (9)
O A. More N2 and H2 would be produced until a new equilibrium
position was reached.
B. More NH3 would be produced until a new equilibrium position was
reached.
O C. The concentrations of N2, H2, and NH3 would decrease until
equilibrium was reached again.
D. More N2, H2, and NH3 would be produced until a new equilibrium
position was reached.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:30, kinggonzales5082
During metamorphic processes, increased pressure and temperature can affect the of minerals in rock. rocks subjected to very high pressure are typically than others because mineral grains are squeezed together, and the atoms are more closely packed. during metamorphic processes, water facilitates the transfer of ions between and within minerals, which can the rate at which metamorphic reactions take place. the growth of new minerals within a rock during metamorphism has been estimated to be about per million years. metamorphism is commonly associated with convergent plate boundaries, where two plates move toward each other. during contact metamorphism, a large intrusion will contain thermal energy and will cool much more slowly than a small one. metamorphosed sandstone is known as the metamorphic rock made from metamorphosed shale, was once used to make blackboards for classrooms.
Answers: 1

Chemistry, 21.06.2019 16:20, maevemboucher78
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2(g)→2alcl3(s) what is the maximum mass of aluminum chloride that can be formed when reacting 32.0 g of aluminum with 37.0 g of chlorine? express your answer to three significant figures and include the appropriate units.
Answers: 2

Do you know the correct answer?
A mixture of reactants and products for the reaction shown below is at
equilibrium in a 5.0 L cont...
Questions in other subjects:

Mathematics, 16.07.2019 21:00

History, 16.07.2019 21:00




Mathematics, 16.07.2019 21:00


Mathematics, 16.07.2019 21:00

Mathematics, 16.07.2019 21:00

Mathematics, 16.07.2019 21:00






