
Assuming that no equilibria other than dissolution are involved, calculate the concentration of all solute species in each of the following solution of salt in contact with a solution containing a common ion. Show that changes in the initial concentrations:
PbCl2(s) in 0.631 M MgCl2 (MgCl2 is strong electrolyte)
Ksp PbCl2 = 1.6 × 10−5

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:30, chelseychew32
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 15:40, alleshia2007
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 16:00, annsmith66
What statement goes against the kinetic theory of gases
Answers: 1

Chemistry, 23.06.2019 00:10, Rubendelarosa1529
Covalent compounds: mastery test select the correct answer what is formed when atoms join together with a covalent bond? a. an ion b. a molecule c. a neutral atom d. a noble gas
Answers: 3
Do you know the correct answer?
Assuming that no equilibria other than dissolution are involved, calculate the concentration of all...
Questions in other subjects:



Mathematics, 30.09.2020 05:01

Physics, 30.09.2020 05:01



Spanish, 30.09.2020 05:01



Social Studies, 30.09.2020 05:01


![[Mg^{2+} ] = 0.631 M \\ [Pb] = 1.0 \times 10^{-5} M \\[Cl^{-} ] = 1.26 M](/tpl/images/1407/4448/37a02.png)
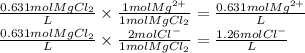
![Ksp = 1.6 \times 10^{-5} = [Pb^{2+} ][Cl^{-} ]^{2} = (x) (1.26+x)^{2}](/tpl/images/1407/4448/5a884.png)
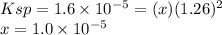
![[Mg^{2+} ] = 0.631 M\\ [Pb] = x = 1.0 \times 10^{-5} M\\[Cl^{-} ] = 1.26+x = 1.26 M](/tpl/images/1407/4448/f1396.png)




