Answers: 1
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 00:40, joe7977
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1

Chemistry, 23.06.2019 10:50, uh8hardiek
Mach the labels with the symbols on the weather map
Answers: 2

Chemistry, 23.06.2019 12:30, Vipain02
The equilibrium constant kc for the reaction 2 nocl(g) → 2 no(g) + cl2(g) is 0.453 at a certain temperature. a mixture of nocl, no, and cl2 with concentrations 1.30, 1.20, and 0.600 m, respectively, was introduced into a container at this temperature. which of the following is true? 1. no apparent reaction takes place. 2. [cl2] = 0.30 m at equilibrium. 3. nocl(g) is produced until equilibrium is reached. 4. [nocl] = [no] = [cl2] at equilibrium. 5. cl2(g) is produced until equilibrium is
Answers: 3
Do you know the correct answer?
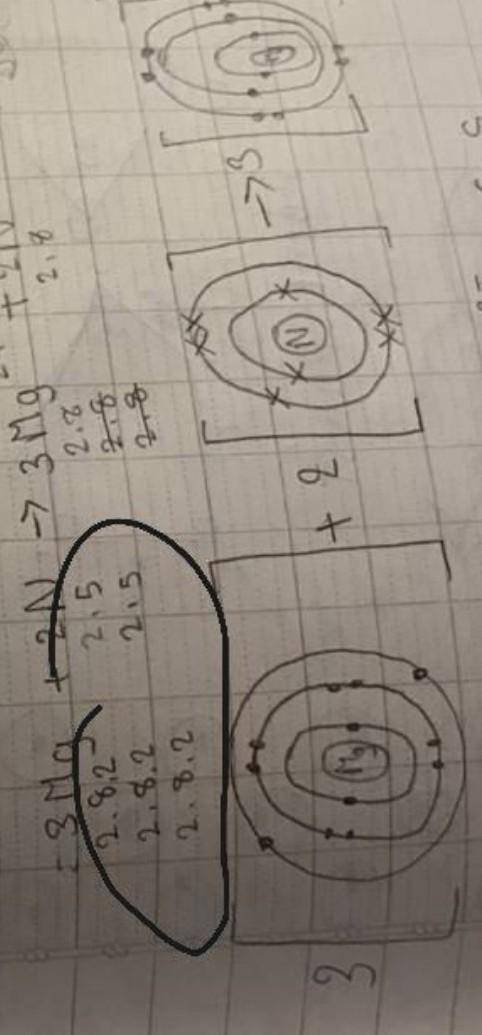
Can someone tell me what does the numbers in the circle mean? and how do I find it?
...
...
Questions in other subjects:


Mathematics, 26.02.2021 06:10

Mathematics, 26.02.2021 06:10


Mathematics, 26.02.2021 06:10

English, 26.02.2021 06:10

Mathematics, 26.02.2021 06:10