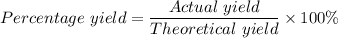Calcium oxide reacts with water in a combination reaction to produce calcium hydroxide: CaO (s) + H2O (l) → Ca(OH)2 (s) In a particular experiment, a 2.00-g sample of CaO is reacted with excess water and 2.14 g of Ca(OH)2 is recovered. What is the percent yield in this experiment? a. 107 b. 1.07 c. 2.88 d. 81.1 e. 93.3

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, paolaviviana
What does earth’s rotation on its axis cause? the tides night and day passing of years phases of the moon
Answers: 1

Chemistry, 22.06.2019 04:30, only1cache
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3


Chemistry, 22.06.2019 12:00, carvajalj2520
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1
Do you know the correct answer?
Calcium oxide reacts with water in a combination reaction to produce calcium hydroxide: CaO (s) + H2...
Questions in other subjects:



Biology, 01.12.2021 05:50





Biology, 01.12.2021 05:50

Mathematics, 01.12.2021 05:50

Physics, 01.12.2021 05:50