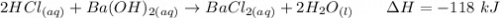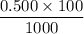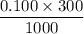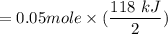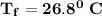Chemistry, 30.07.2021 23:00, maskythegamer
Calculate the heat when 100.0 mL of 0.500 M HCl is mixed with 300.0 mL of 0.100 M Ba(OH)2. Assuming that the temperature of both solutions was initially 25.0C and that the final mixture has a mass of 400.0 g and a specific heat capacity of 4.18 J/C g, calculate the final temperature of the mixture.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:00, melissa9882
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

Chemistry, 22.06.2019 13:30, annanikherrera
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 22.06.2019 15:20, mydoggy152
Fossil fuels are organic compounds that are made from
Answers: 1
Do you know the correct answer?
Calculate the heat when 100.0 mL of 0.500 M HCl is mixed with 300.0 mL of 0.100 M Ba(OH)2. Assuming...
Questions in other subjects:




Mathematics, 13.09.2021 21:20



Health, 13.09.2021 21:20


Health, 13.09.2021 21:20