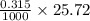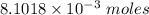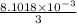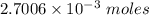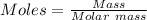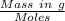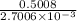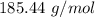Chemistry, 30.07.2021 19:50, elmoreroman11
0.5008 g of an unknown triprotic acid, H3A, is dissolved in 47.3 mL of water and then titrated with 0.315 M NaOH. It takes 25.72 mL of the NaOH solution to completely neutralize the acid. What is the molar mass of this acid

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:00, aschool2000
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2


Chemistry, 22.06.2019 14:00, MathChic68
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1
Do you know the correct answer?
0.5008 g of an unknown triprotic acid, H3A, is dissolved in 47.3 mL of water and then titrated with...
Questions in other subjects:


Mathematics, 16.02.2021 19:10

Mathematics, 16.02.2021 19:10

Mathematics, 16.02.2021 19:10

Mathematics, 16.02.2021 19:10


Mathematics, 16.02.2021 19:10

Mathematics, 16.02.2021 19:10