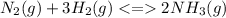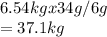Chemistry, 27.07.2021 02:00, dbzrules02
A reaction vessel for synthesizing ammonia by reacting nitrogen and hydrogen is charged with 6.54 kg of H2 and excess N2. A total of 30.4 kg of NH3 are produced. What is the percent yield of the reaction

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, giraffegurl
If 34.2 grams of lithium react with excess water, how many liters of hydrogen gas can be produced at 299 kelvin and 1.21 atmospheres? 2 li (s) + 2 h2o (l) yields 2 lioh (aq) + h2 (g)
Answers: 3


Chemistry, 22.06.2019 16:50, briansalazar17
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3
Do you know the correct answer?
A reaction vessel for synthesizing ammonia by reacting nitrogen and hydrogen is charged with 6.54 kg...
Questions in other subjects:

Mathematics, 20.05.2020 04:00

History, 20.05.2020 04:00

Biology, 20.05.2020 04:00

Mathematics, 20.05.2020 04:00

English, 20.05.2020 04:00

Mathematics, 20.05.2020 04:00

History, 20.05.2020 04:00

Mathematics, 20.05.2020 04:00

Mathematics, 20.05.2020 04:00

History, 20.05.2020 04:00