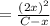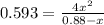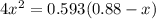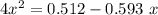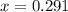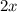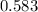Chemistry, 25.07.2021 20:20, marissalwilliams3
For the following reaction of N2O4, the equilibrium constant is 0.593 at a particular temperature.
N2O4(g) ⇌ 2 NO2(g)
If the initial concentration of N2O4 is 0.880M, what are the equilibrium concentrations?
Please show work!

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:00, queenkimm26
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 22.06.2019 18:00, jalenclarke25
What volume would 2.25 moles of ne has occupy at stp?
Answers: 1
Do you know the correct answer?
For the following reaction of N2O4, the equilibrium constant is 0.593 at a particular temperature....
Questions in other subjects:

Biology, 09.11.2020 21:40

Mathematics, 09.11.2020 21:40

Physics, 09.11.2020 21:40

Mathematics, 09.11.2020 21:40





Mathematics, 09.11.2020 21:40

English, 09.11.2020 21:40


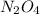 be "C".
be "C".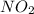 be "x".
be "x".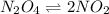
![K_c = \frac{[NO_2]^2}{[N_2O_4]}](/tpl/images/1399/4499/98df0.png)